
Check Out Our New Forum!
The Your Turn Forum provides a space where people can post questions about mesh and medical device related issues and get advice and support from the Mesh News Desk community.
Join the Discussion!
TVT Secur, Megamed Service, RU
October 27, 2015 ~ The Josephine Marie Rabiola v. Ethicon trial, scheduled for Monday, October 26 in Austin, Texas has been rescheduled to February 2016 at the Austin court's request. The judge is not set in this rotating docket. The case is expected to be rescheduled one more time at a later date, perhaps mid-2016.
Rabiola is represented by the Matthews Law Firm as well as Freese and Goss.
The case was remanded from multidistrict litigation in Charleston, WV to Austin, Texas - Josephine Marie Rabiola v. Ethicon, 53rd Judicial District Court, Austin TX. Case no. D-1-GN-13-002039.
Ms Rabiola, of Austin, Texas, was 63 years old when she was implanted with two meshes - first the Gynecare TVT Secur System to treat stress urinary incontinence (SUI) on May 24, 2010. She was then implanted with Gynecare Prosima Pelvic Floor System to treat pelvic organ prolapse (POP) on November 2, 2011.

TVT Secur tip, Neuman Urology, Israel
The meshes are made by Ethicon, a division of Johnson & Johnson and include polypropylene mesh tethered in place with two arms that extend up through the buttocks. They were approved through the FDA’s 510(k) approval process.
Both meshes were removed from the market in 2012. See the Mesh News Desk story here.
Even though they were voluntarily removed, a quick online search fines one can purchase the TVT-Secur for a suggested price of $900 here. (No date of this post is given.)
A legal victory was just handed to Ethicon this month in the Dallas trial of Cavness v. Ethicon. She was implanted with the Prosima mesh for pelvic floor repair.
Rabiola has included her implanting doctors in the action - Tomas G. Antonini M.D. of Lone Star Urogynecology and Continence Center, Seton Healthcare Family of Hospitals including its Medical Center, Johnson & Johnson and Ethicon.
THE COMPLAINT
The complaint says despite the claims polypropylene mesh is inert, scientific evidence shows the material is “biologically incompatible with human tissue and promotes an immune response.” In addition, it degrades, causing severe adverse reactions to the mesh including shrinkage from 30-50%, erosion and bacteria harbored in the small weave. Polypropylene is impure, the complaint says.

“There is no such thing as pure polypropylene. Polypropylene contains about 15 additional compounds which are leached from the polypropylene and are toxic to tissue which enhances the inflammatory reaction and the intensity of fibrosis.”
Mesh anchors are likely to pass through and injure major nerve routes in the pelvic region. Welding of the mesh during its production creates a toxic substance that contributes to the mesh degradation, says the complaint.
These mesh products were marketed to the public through aggressive campaigns despite the high failure and complication rates, according to attorneys for the Plaintiff who add that J&J and Ethicon have consistently under-reported and withheld information about the propensity of its products to fail and cause injury.
Injuries include mesh erosion, mesh contraction, infection, fistula, inflammation, scar tissue, organ perforation, dyspareunia, blood loss, acute and chronic nerve damage and pain, pudendal nerve damage, pelvic floor damage, chronic pelvic pain, urinary and fecal incontinence and prolapse of organs. Many women must undergo repeat surgeries, pain control, injections, explant surgeries and repair surgeries in an attempt to rebuild what has been damaged.
She suffers physical pain and mental anguish, now and likely in the future; disfigurement; physical impairment and medical expenses, both now and in the future.

Tomas Antonini, M.D.
Ms. Rabiola was implanted on May 24, 2010 by Dr. Chris Hart with the TVT Secur to treat stress urinary incontinence. Dr. Antonini owns the medical group and he knew or should have known the pelvic mesh products had a high failure and injury rate, failed to perform as intended, and often required additional surgeries.
Ms. Rabiola went to Dr. Antonini to consult on the TVT Secur System and he recommended the Gynecare Prosima Pelvic Floor Repair System be implanted. He failed to inform her of all of the risks involved, according to her complaint. Had she known, she would not have consented to the implantations. On November 2, 2011, she had the products implanted to treat SUI and POP.
CAUSE OF ACTION
The causes of action include Negligence- J&J and Ethicon failed to use reasonable care in designing, manufacturing, marketing labeling and packaging its pelvic mesh products.
The doctor and hospital failed to select the proper treatment for SUI and POP patients and improperly selected her as a candidate for such products.
Other cause include Strict Liability, Design Defect – Dr. Hart and Antonini were in the business of supplying these products, which were defectively designed when they were sold and were unreasonably dangerous. The Plaintiff was not warned about the risks. This was proximate cause the damage and injuries to the Plaintiff.
Breach of Implied Warranty – The Plaintiff relied on the implied warranty of merchantability of the Defendant, which they breached because the products were not suited for their intended use.
Breach of Express Warranty – The Defendant made assurances to the public, to hospitals and doctors about the safety and fitness of their pelvic mesh products. Ms. Rabiola relied on those promises.
BACKGROUND ON TVT-SECUR
This will be the first trial of the TVT-Secur System, a mini-sling that mimics the TVT-Obturator placement. See European Urology
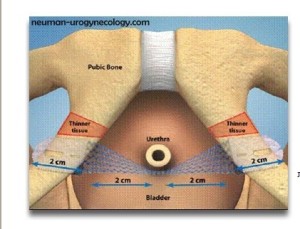
The only other trial of the TVT-Secur involved the case of Stacey Wilkins which was settled in a Joplin, Missouri State Court in May 2014 for an undisclosed amount. Adam Slater was her attorney. See the Mesh News Desk story here.
The TVT-Secur was intended to be the third generation and an improvement on TVT (transvaginal tape). In the case of a similar product, the Abbrevo mini-sling, Plaintiff Coleen Perry received a $5.7 million jury award in March 2015. #